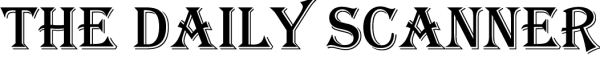Clinical trials are fundamental to medical progress. These carefully designed studies are essential for deepening one’s understanding of diseases and developing effective treatments. However, the entire landscape of clinical research has been transformed with the integration of technology. From patient recruitment to data analysis, technology has reshaped the process, making it more efficient, accurate, and expansive. This article will discuss how this fusion has become a powerful partnership for progress.
- Digital Recruitment Techniques
Historically, the recruitment of participants for clinical trials was a challenge. Traditional methods like newspaper ads or flyers were not only time-consuming but also limited in reach. Enter the age of technology, and platforms like electronic health records (EHRs) now play a transformative role. With EHRs, researchers can rapidly identify potential participants based on health metrics, thereby streamlining the recruitment process.
Moreover, social media platforms and mobile apps have opened new avenues for recruitment. Digital advertising allows for targeted campaigns, reaching potential participants based on demographics, interests, and health conditions. The efficiency and reach provided by these tech tools have substantially reduced recruitment timelines.
- EDC Systems And Automated Metadata Management
Modern clinical trials often employ Electronic Data Capture systems, replacing traditional paper-based methods. Alongside these EDC systems, automated metadata management becomes crucial. It ensures organized, consistent, and easily retrievable data annotations. As researchers input, store, and manage patient data in digital format, metadata management provides an efficient framework to categorize and understand this influx of information.
Not only does this facilitate easier data management, but it also reduces the risk of human errors associated with manual data entry and annotation. With features like automated data validation checks, EDCs and metadata management tools work in tandem to ensure that the captured data remains consistent and accurate throughout the trial.
- Real-Time Data With Wearables
The emergence of wearable devices has been a game-changer for clinical trials. Devices like smartwatches, fitness trackers, and health monitors provide continuous data collection, allowing researchers to gather real-time information on a patient’s vitals, movement, and even sleep patterns.
This continuous data stream offers a more comprehensive understanding of a treatment’s effects over time. Plus, these devices help reduce the need for frequent clinic visits, making participation more convenient for volunteers and reducing overhead costs.
- Embracing Virtual Clinical Trials
Virtual clinical trials, sometimes termed “decentralized trials,” represent another significant stride. These trials use digital tools and platforms to interact with participants, reducing or even eliminating the need for physical site visits. From video consultations with trial coordinators to e-consent forms, technology ensures that trials are both participant-friendly and adaptable to various conditions.
Such trials are particularly beneficial in reaching populations that might be geographically distant or unable to frequent a research facility. They hold the potential to make clinical trials more inclusive and diverse, ensuring treatments are effective for a broader range of patients.
- AI In Data Analysis
Handling and analyzing the vast amount of data generated in a clinical trial can be daunting. AI and machine learning algorithms offer a solution. These tools can rapidly sift through data, identifying patterns, anomalies, or correlations that might be overlooked in manual analysis.

This not only speeds up the research process but also potentially uncovers novel insights. Predictive analytics, a branch of AI, can even anticipate outcomes based on initial data, guiding researchers in making informed decisions as trials progress.
- Blockchain For Data Integrity
Data integrity is paramount in clinical trials. Any discrepancies or tampering can jeopardize the validity of the entire study. Blockchain technology offers an immutable and time-stamped record of data entries. Once a piece of information is added to the blockchain, it can’t be altered without leaving a trace.
This ensures that data remains untouched and verifiable, fostering trust in the results. Moreover, blockchain’s decentralized nature ensures that multiple stakeholders can access the data simultaneously, promoting transparency in the process.
- The Role Of Telemedicine
Telemedicine platforms have transformed patient-researcher interactions. These platforms allow for remote consultations, enabling researchers to monitor patients, address concerns, and ensure adherence to trial protocols. During situations like global pandemics, telemedicine ensures that clinical trials can continue without significant interruptions, safeguarding both the progress of research and the health of participants.
- Mobile Apps For Patient Adherence
Patient adherence to trial protocols is essential for valid results. Mobile applications, with reminders, informational materials, and communication tools, ensure that participants are well-informed and engaged throughout the trial. By providing a convenient platform for participants to log their experiences, report side effects, or communicate concerns, these apps facilitate better trial outcomes and patient experiences.
- Direct Feedback With ePRO
Electronic Patient-Reported Outcomes (ePRO) platforms allow participants to provide feedback directly, often through a tablet or mobile application. This direct method eliminates the need for intermediaries and minimizes data entry errors. It ensures that patient experiences, symptoms, and side effects are documented in real-time, providing a more accurate representation of the treatment’s impact.
- Genomics And Personalized Medicine
Technological advancements in genomic sequencing have paved the way for personalized medicine within clinical trials. By analyzing a participant’s genetic makeup, researchers can predict how individuals might respond to a particular treatment. This level of precision ensures that patients receive treatments tailored to their genetic profile, maximizing efficacy and minimizing potential side effects.
- Advanced Imaging For Deeper Insights
With the advent of sophisticated imaging technologies like functional MRI and PET scans, researchers can delve deeper into the physiological effects of treatments. These technologies offer visual insights into cellular and molecular processes, allowing for a better understanding of how a drug or treatment modulates disease processes.
- Cloud Technology In Collaboration
Cloud technology has been instrumental in bridging the gap between multiple stakeholders in a clinical trial. Researchers, sponsors, CROs (Clinical Research Organizations), and other involved parties can access trial data from anywhere in the world, fostering a collaborative environment. Moreover, cloud platforms offer scalable storage solutions, accommodating the vast volumes of data generated during trials.
Wrapping Up
The intersection of clinical trials and technology signifies a promising horizon for medical research. With technological tools addressing historical challenges and opening new possibilities, the efficiency and scope of trials have expanded significantly. These advancements not only promise quicker turnarounds in research but also ensure that clinical trials are more participant-centered, transparent, and globally inclusive.